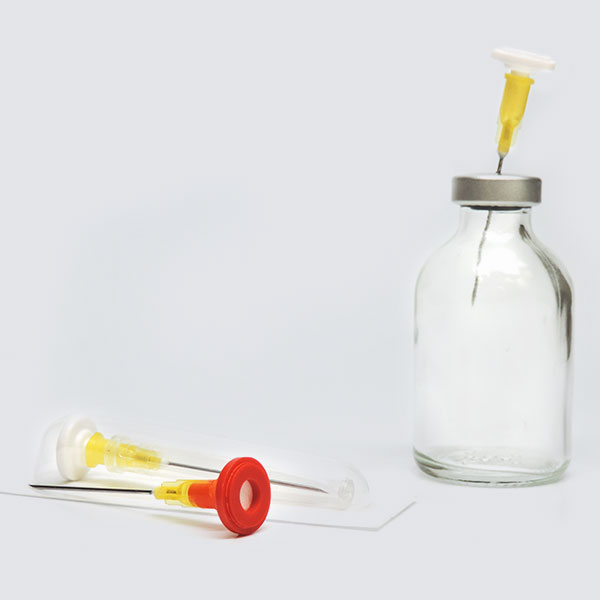
Trusted Security. When you need it most.
Solutions to Enhance Drug Safety
When your vaccine protocols call for pre-filled syringes, you can trust IMI to ensure the integrity of your preparations, whether therapeutics or vaccines. Over 80% of the leading US Outsourced Compounders trust Prep-Lock™ Tamper Evident Caps to provide an additional level of security to their IV Syringe Medications. The United States Pharmacopeia (USP) states the use of tamper-evident closures on the ports of compounded sterile preparations can add an additional level of security to ensure product integrity. Prep-Lock™ Caps greatly reduce the risk of your CSPs being compromised and ensure product integrity is maintained from the time it leaves the pharmacy until administered by an authorized clinician.

“The Royale Pharmaceutical Society recommends that to minimize manipulation of medicines in clinical areas injectable medicines should be in prefilled syringes (PFS) wherever possible.”
Increase Sterility & Reduce Contact Time
A recent article published in The BMJ suggests having injectable medications in prefilled syringes, whenever possible, could greatly improve the mass vaccination processes. When protocols do not require injectables to be prepared at the point of administration, fewer skills and less training are required. As a result “the process speeded up, probably reducing patient vaccinator contact time.” Additional studies support the use of prefilled syringes to reduce the time of infusion and risk of infection. In addition to expediting processes, The BMJ states PFSs will make the process more sterile. “Some of the side-effects such as redness and soreness at the injection site may be due to inadvertent contamination during the normal syringe preparation process, reported as high as 6%.”
Download The Facts Sheet
Support Mass Vaccination
Reduce contact time and increase the number of qualified administrators.
Increase Sterility
Reduce side-effects such as soreness of injection site by avoiding inadvertent contamination.
Reduce Transmission
The use of multidose vaccine vials presents the potential for unintended hepatitis transmission.
USP <797> Standard Security
The purpose of USP Chapter <797> is to ensure the safety and protection of patients and healthcare workers involved in sterile compounding preparations by providing a set of enforceable standards that describe the conditions and practices to reduce the potential of microbial contamination. The chapter presents minimum practices and quality standards for Compounded Sterile Preparations CSPs based on the most recent scientific information and best sterile compounding practices. “The use of tamper-evident closures and seals on CSP ports can add an additional measure of security to ensure product integrity regardless of the transport method used.”
RELIABLE QUALITY & SECURITY
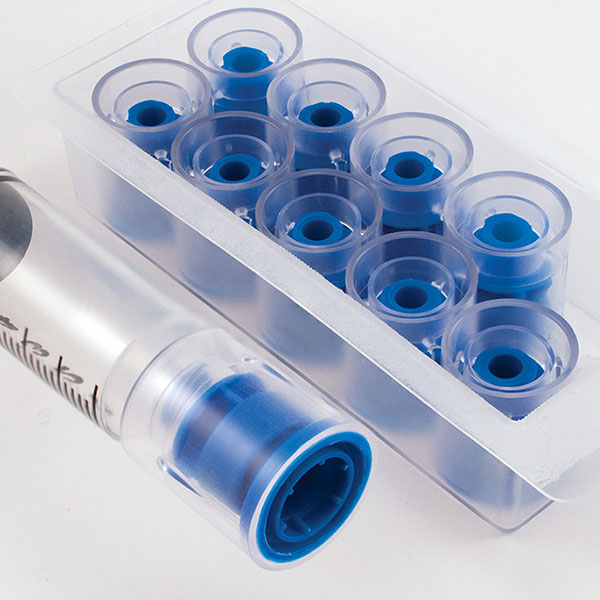
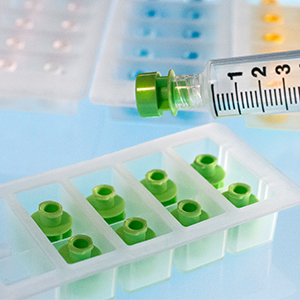
Each sterile tray of 10 caps offers the pharmacist the ability to quickly install the cap with a simple twist of the syringe. This helps to reduce the risk of touch contamination and enhances aseptic technique. Administering a syringe is very easy for the authorized clinician. Simply pull-off the outer sleeve of the tamper-evident cap, unscrew the remaining Luer-lock cap, and discard.
A choice of three different colored caps and two outer sleeve styles allows the pharmacy the ability to color-code either specific drug classes or processes.
Download Product Data Sheet
Download Instructions For Use
Contact Us
Have questions about what a partnership with IMI can do for your organization? What more information on a product or pricing? Fill out the form below and one of our Specialists will get back to you as soon as possible (usually within one business day) or call the number to the right.
We offer complementary evaluatory samples.
(800) 344-2554
You May Also Be Interested In
IMI remains at the forefront of developing new products specifically designed for the compounding industry. Our strategic partnerships with some of the biggest names in health care enable us to continusoulsy deliver the kind of high quality and high-value products that pharmacy operations demand. All IMI products are manufactured in the United States under the strictest quality standards at our FDA-registered, ISO 13485–certified facility. Browse our Comprehensive product families below, and as always our premium products are back with premium service. If you have any questions, contact us, we’re here to help!