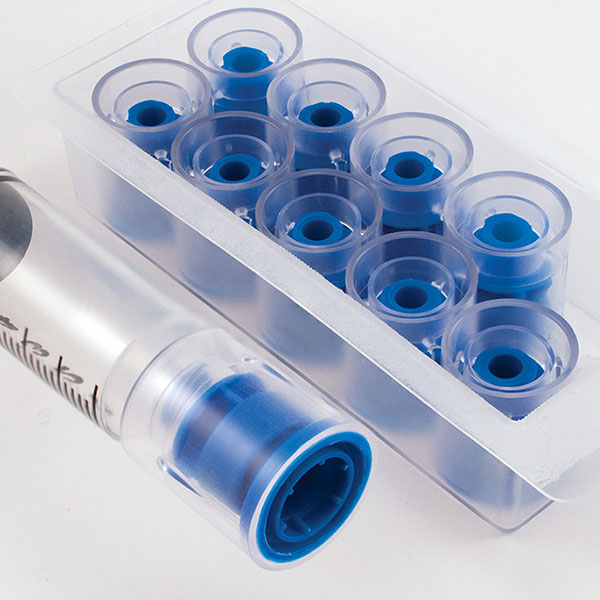
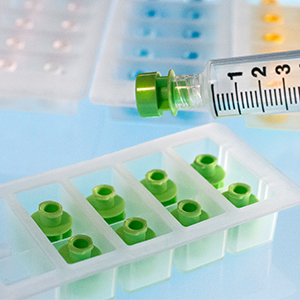
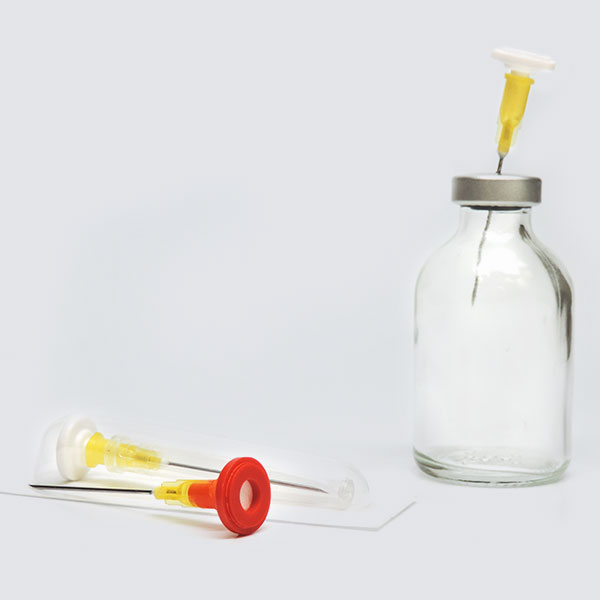
Prep-Lock™
Tamper Evident Caps
For Oral Syringes
SIMPLE SOLUTIONS FOR GUARDING ORAL SYRINGES.
For over 50 years, IMI has delivered high-quality, American made devices for the pharmacy. By building long-standing relationships with pharmaceutical and healthcare professionals, we continue to advance our devices to best serve the healthcare industry. Prep-Lock™ Tamper Evident Caps for Oral Syringes are a prime example of our customer lead ethos. Using IMI’s patented breakaway sleeve technology, each cap is designed with unique specifications compatible with Baxter Exacta-Med™ and BD UniVia™ Oral Syringes.

Deterrent to Diversion
Serves as an active deterrent to potential drug diversion and misuse
Industry Leading
Through 50 years of service, IMI has become the standard for guarding Compounded Sterile Preparations.
DEA & USP
Tamper evident protocols may help your pharmacy comply with DEA and USP guidelines.
Feature Enhancements
Tamper Evident Caps for Oral Syringes now Featuring Retained Indicator Ring
- Retained Ring feature for improved control of disposable components.
- Retained Ring feature compatible with Baxter Exacta-Med™, BD Univia™, and Comar™ Oral Syringes.
- Tampering is immediately evident when the colored outer sleeve is broken or missing.
- Increased level of confidence for healthcare practitioners.
- Tamper-evident protocols may help your pharmacy copy with DEA & USP guidelines.
Download Instructions for Use >
ADVANCING THE SAFETY AND SECURITY OF YOUR DRUG PRODUCTS AND ULTIMATELY YOUR PATIENTS
Oral syringe administration is often required to meet the medication needs of neonatal patients, children, and adults who are unable to swallow solid dosages. Prep-Lock Tamper Evident Caps are the industry standard for safeguarding compounded preparation from the time they leave the pharmacy until an authorized clinician administers them.
Drug diversion concerns are driving the need to secure and protect syringe contents. Tamper Evident Caps provide an active deterrent to diversion and misuse. Securing syringes with Tamper Evident Caps helps to ensure that medications arrive to patients uncompromised and makes a clear statement about how seriously your operation takes drug security.
Evaluation Samples Available
If you have questions regarding what a partnership with IMI could do for your organization, or you would like more information on a product, pricing, or requesting free samples please fill out the form below. One of our specialists will get back to you as soon as possible (usually within one business day).
You May Also Be Interested In
IMI remains at the forefront of developing new products specifically designed for the compounding industry. Our strategic partnerships with some of the biggest names in health care enable us to continusoulsy deliver the kind of high quality and high-value products that pharmacy operations demand. All IMI products are manufactured in the United States under the strictest quality standards at our FDA-registered, ISO 13485–certified facility. Browse our comprehensive product families below, and as always our premium products are back with premium service. If you have any questions, contact us we’re here to help!