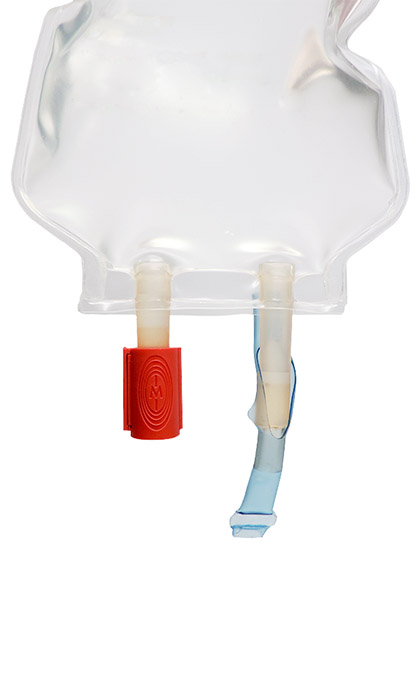
Tamper Evident Additive Port Cap
IMI has recently released two new products into their Prep-Lock™ Family of Products. IMI’s Prep-Lock product line offers a variety of ways to enhance the security of your compound sterile preparation from the moment it leaves the sterile hood until administered by an authorized clinician. The innovative devices offered within the Prep-Lock product line are an important component of any healthcare system’s drug security program.

An essential component of a Health System’s overall drug security program
The Tamper Evident Additive Port Cap for use with Baxter Viaflex, Intravia, Aviva, and All-in-One containers offers protection against potential tampering or misuse of your preparation. The design allows for one-handed installation onto the additive port and can also serve as an indicator that medication has been added. Tamper evident technology ensures that once closed, the Additive Port Cap cannot be removed without damaging the IV bag.
Related Articles
Bryan McGurn, Sr. VP of Sales & Marketing Discusses IMI’s Impact In The Healthcare Domain
Bryan McGurn, Sr. VP of Sales & Marketing Discusses IMI's Impact In The Healthcare DomainEvaluating IMI's contributions to healthcare systems and specialty pharmacies alike through highly valued partnerships, advocating for customers' voices, and adapting to new...
International Medical Industries Releases Tamper Evident Caps with Incorporated RFID
IMI (International Medical Industries, Inc.) bolsters expertise in secure drug delivery products with the release of their Prep-Lock Tamper Evident Cap for IV Syringes with incorporated Radio Frequency Identification (RFID) technology.
Tamper-Evident Caps with RFID Combat Diversion in Healthcare
The Staggering Cost of DiversionA recent study published in the Journal of Hospital Medicine cited the vast costs diversion poses for Health Systems. “Hospitals bear the cost of diverted drugs, internal investigations, and follow-up care for affected patients, and can...
